ABS-M30i
FDM Thermoplastic
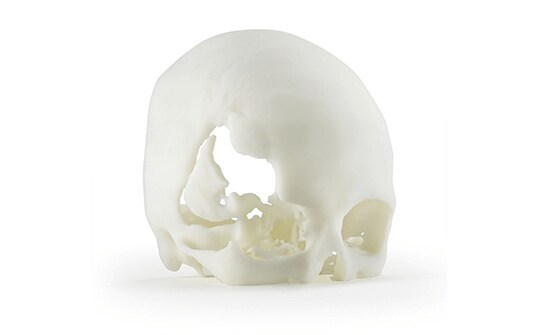
ABS-M30i is a biocompatible 3D printing material perfectly suited for medical, pharmaceutical and food-packaging engineers and designers to produce surgical planning models, medical devices, tools and fixtures.
High Strength, Sterilizable FDM Material
ABS-M30i is a strong, biocompatible material capable of sterilization and suitable for use in medical devices. It complies with the test requirements of ISO 10993 (biological evaluation of medical devices), USP Class VI (plastic biocompatibility) and ISO 18562 (biocompatibility of breathing gas pathways in applications such as ventilators, breathing tubes, resuscitators and other airway-related devices). Test results for ISO 18562 are available from the link below.
Performance properties
TENSILE STRENGTH
31 MPa (XZ Axis), 26 MPa (ZX Axis)
TENSILE MODULUS
2,180 MPa
HDT @ 66 PSI
96 °C
IMPACT TOUGHNESS
IZOD Impact, notched – 2.4 ft-lb/n (XZ Axis), 128 J/m (ZX Axis)
Compatible printers


Fortus 450



